(A) A pair of dice is rolled one time, what is the probability.Consider reaction (1) shown below with it's associatedĮquilibrium constant.
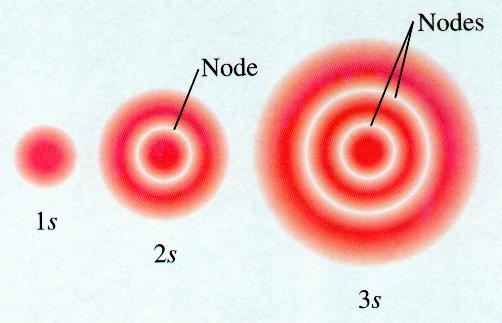
#Two types of nodes occur in atomic orbitals free
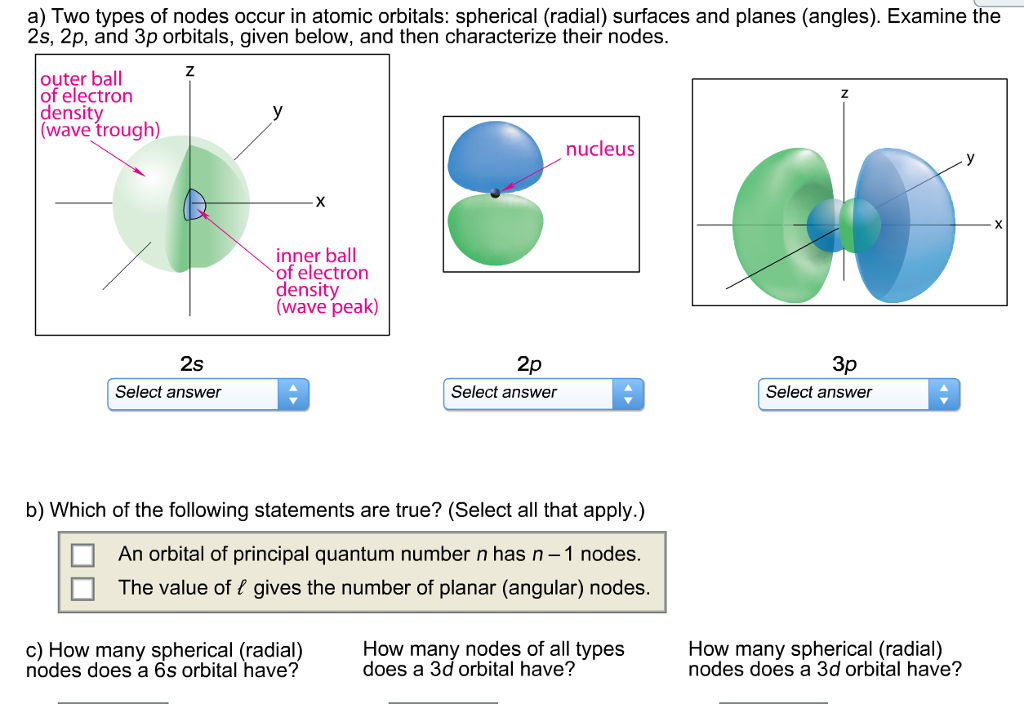
?��⃗ = 3?�?+√2?�? is incident from free space onto aĭielectric material with ε=2εo as below.Nodes are classified into two types: radial nodes and angular nodes. Is your hair curly, straight, or wavy? In humans, straight hair The nodal point is a point where there is no chance of finding the electron.Providing examples from your daily life that illustrates. Explain the terms “deductive reasoning” and “decision making”,.Fairmount Travel Gear produces backpacks and sells them to.The ac adapter for a laptop computer contains a transformer. Consider a LTI system with impulse response h = u*a^n,.O2 has two unpaired electrons in its orbitals, and a bond order of 2. QUESTION 11 Match the alom indicated with the correct hybridization for the molecule CHCOOH (scetic acid) of the CH csup>3 - Cof the COOH - In the CEO bond - in the CO H portion Acis in CO bond and thus is sp Bo has electron domains but is sp C carbon has & electron domains and so is sp 0.has only 3 electron domains and so is sp? QUESTION 12 in the compound CH CH The hybridization of both. The Lewis structures have an unpaired electron and an average bond order of 1.5. QUESTION 11 Match the alom indicated with the correct hybridization for the molecule CHCOOH (scetic acid).Which one of the following elements forms a stable 2- anion? (1) P (2) Se (3) AI (4) Mg 21. Which element has the following ground-state electron configuration (Kry5s4d105p? (3) 1 (4) Te (1) Sn (2) Sb 20. When the atomic orbitals of the two atoms combine, the electrons occupy the molecular orbital of. How many electrons must a Sc atom lose to form a stable cation? (1) 5 (2)3 (3)7 (4) 8 19. A dihydrogen molecule (H2) forms from two hydrogen atoms. How many valence electrons does Br atom have? (1)2 (2) 4 (3) 7 (4) 8 18. Examine the nodes in 2s,2p, and 3p orbitals, and show that they agree with. How many valence electrons does Br atom have? (1)2 (2) 4 (3) 7 (4) 8.ħ. (a) Two types of nodes occur in atomic orbitals: spherical surfaces and planes. The two colors show the phase or sign of the wave function in each region. Penalties are applied for incorrect guesses. The shapes of the first five atomic orbitals are: 1s, 2s, 2px, 2py, and 2pz. What are the possible values of l? Select all that Hybridization is a model used to describe the observed bonding in molecules. ( here is circle cant draw it)ī) If the n quantum number of an atomic orbital is 1, An atom can have both hybridized and unhybridized orbitals at the same time. Note: There are no radial nodes in this orbital. 1 a)The picture below best represents which atomic orbital?ġ a)The picture below best represents which atomic orbital?.
